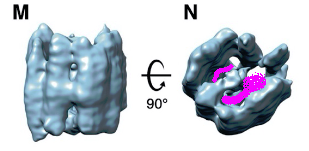Antiviral drugs specific for coronaviruses in preclinical development
https://pdf.sciencedirectassets.com/270558/1-s2.0-S1879625714X00027/1-s2.0-S1879625714001370/main.pd…
It’s mutating faster than expected with implications for leathality.
Patient-derived mutations impact pathogenicity ofSARS-CoV-2
https://www.medrxiv.org/content/10.1101/2020.04.14.20060160v1.full.pdf
Nucleoproteins and spiders web
Eli I wonder if the regularity of the nucleoproteins around the viral RNA can be due to a similar mechanism, that what happens when water droplets spread out in regular intervals on a spider’s web. So that it is favorable energywise for the water to arrange around the nucleoproteins, if they are placed regularly.
Image by PublicDomainPictures
I found a fine explanation of the physics behind.
What makes water-drops/dew stick on a spider’s web and what keeps them there?
I know the nucleoproteins bind to the RNA due to it being positively charged and RNA being negatively charged.
By nucleoproteins binding to the RNA, I think it will have to replace some water. What if the lowest energy of placement is in a regular fashion?
Otherwise the octamers of nucleoproteins look like they can attach to each other.
More water can be packed around the nucleoproteins than could be packed around the RNA itself.
rhiju das This is a beautiful idea. Have you heard about recent hypotheses regarding RNA-protein condensates and liquid-liquid phase separation in biology?
Eli No. I have today read about condensation in relation to spiderwebs, and a wet spider web being more elastic and stronger.
Please do throw me a paper ![]()
“My” paper is called Wet webs work better: Humidity, super contractions and the performance of spider orb webs.
Wet webs work better: humidity, supercontraction and the performance of spider orb webs
RNA with nucleoproteins around them are kind of an RNA/protein hybrid. So I wouldn’t be surprised if it goes towards having sort of a protein like character. Since the nucleoproteins seems to be what there is most of, when it comes to amount and weight.
Eli rhiju, this is fascinating. I looked up two major ampullate silk proteins. They have crazy amino acid repeats. I knew as I had looked at spider silk proteins before. But when I check a sars nucleoprotein, (I’m not aware which is the right one to look at, so I just grabbed a random one) it does not have nearly the same amount of repeat, but it has some. It also carries a heavy amount of Q, something spider silk protein does to.
https://www.ncbi.nlm.nih.gov/protein/AFN54362.1?report=fasta
https://www.ncbi.nlm.nih.gov/protein/AGT20893.1?report=fasta
Ok, I see that Q is also scattered well out over other proteins. Anyway, I think it looks like the sars nucleoprotein has a bit more amino acid repeats, than what I see in general for average proteins.
rhiju das Look for disordered domains of the protein. (Not in spider silk, in nucleoprotein). In human proteins tails and disordered domains are increasingly found to mediate formation of gels or liquid-like condensates if proteins that associate with RNA. Try to find a review of stress granules or P granules or the nucleolus - May lead you to new hypotheses
Eli How would I know what is the disordered part of the protein?
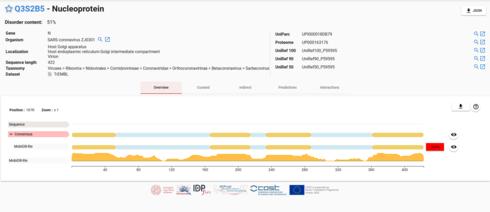
Cool, I found a manually curated database over disordered proteins:
Beginning and end are judged to be disordered, but the middle is ambiguous.
I found something better:
The yellow area are called polyampholyte
https://www.advancedsciencenews.com/polyampholytes-for-biomedical-applications/
Also the repeats amino acids that stood out to me, are specifically in the middle and last yellow region.
When I look at the nucleoprotein in PDB, it makes sense that the ends and the middle are unstructured.
PDB nucleoprotein sars
The protein is split in two and there is a linker in the middle.
http://www.rcsb.org/3d-view/3I6L
rhiju das here’s a paper on this new concept of RNA-protein condensation
https://www.nature.com/articles/nrm.2017.7
(the full paper is available as a pdf, if you search the web.)
Eli Human histones do not to nearly the same degree seem to have these disordered regions as the corona nucleoprotein. (Except for the H5 linker histone) So perhaps this paper may explain why the corona nucleoproteins does not jump on cellular DNA or RNA, because they may be in separate regions of the cell. But histone tails are still highly modifiable while not deemed disordered.
The nucleoproteins are separated from the M, E and S protein in its processing. I’m not sure though where in the cell it is estimated to be.
I have been watching an Ibiology video on histones to grasp the topic better. HATs is histone acetyltransferases that modifies the lysines in the tail of some histones h2B, H3 and H4 (https://en.wikipedia.org/wiki/Histone_acetyltransferase)
Liquid Phase Separation in Living Cells by Cliff Brangwynne
This whole IBiology series of 3 are excellent. They explain well lipid-lipid phase separation. In short, it gets explained why cells are not just one big mess of things swimming around, but there are seperate comparments. Also outside of organelles.
Eli Speaking of stress granules. I earlier saw a paper that went on a fishing expedition in the human genome with the corona virus proteins, to see what interacted. I was wanting to know if HAT or HDAC was interacting with Nucleoproteins. I didn’t get lucky on that one. Although HDAC2 is involved somehow.
https://www.biorxiv.org/content/10.1101/2020.03.22.002386v1
Histone deacetylases (EC 3.5.1.98, HDAC) are a class of enzymes that remove acetyl groups (O=C-CH3) from an ε-N-acetyl lysine amino acid on a histone, allowing the histones to wrap the DNA more tightly. https://en.wikipedia.org/wiki/Histone_deacetylase
I am still interested in acetylation and deacetylation in relation to the nucleoprotein. An experiment I would love to see done is:
-
Corona RNA and loose nucleoproteins + Histone deacetylases. Guess packing
-
Packaged corona RNA wrapped in nucleoproteins + Histone acetyltransferases. Guess, unpackaging
I realize that this scientist team fished with single proteins alone. They did not fish with the viral RNA in combination with the nucleoproteins. So they can say nothing about if there is an interaction between nucleoproteins and deacetylases/acetyltransferases. This means that RNA histones are still on the table.
rhiju das check out relevant new preprint!
https://www.biorxiv.org/content/10.1101/2020.04.02.022194v1
Eli Thx also for the Nucleoprotein paper. I like how the story goes with your addition of the liquid-liquid phase separation in relation to RNA and proteins. I have read the paper you shared, watched a couple of Ibiology videos on the topic and read a few more papers. I will try to drag out what I think is relevant to our story.
The genomic corona RNA and the nucleoproteins can be in a sort of p-body. As far as I understand coronavirus replicates in the cytoplasm and not the nucleus.
The viral RNA and nucleoproteins being together in small droplet compartments could explain the why of your question of why the nucleoproteins do not attach to host mRNA.
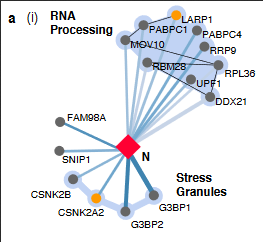
This made me wonder if it is known if coronavirus or viruses in general replicates in p granules or stress granules.
“Like that of other positive-strand RNA viruses, coronavirus RNA synthesis is associated with extensively rearranged intracellular membranes (38). High-resolution three-dimensional images obtained by electron tomography in SARS-CoV-infected cells showed a unique reticulovesicular network of modified endoplasmic reticulum that integrated convoluted membranes (CMs), interconnected double-membrane vesicles (DMVs), and vesicle packets apparently arising from DMV merger.”
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6025776/
Which reminds me that I love all those scientists who kept working on SARS when there were no current threats.
"Stress granules and processing (P) bodies are cytoplasmic RNA granules that contain translationally silenced messenger ribonucleoproteins, contributing to translation regulation in cells. Whereas P bodies are constitutively expressed and include components involved in mRNA decay, stress granules are thought to be sites of mRNA storage and triage formed in response to stress conditions. Stress granules represent an intermediate stage in the dynamic equilibrium between active translation on free polysomes and mRNA decay in P bodies (53, 54).
During infection, RNA viruses dynamically interact with stress granules and P bodies (55), leading to varying stress granule phenotypes. Many viruses have evolved mechanisms to antagonize the formation of stress granules, suggesting that stress granules are involved in restricting virus replication through RNA silencing (56, 57). In contrast, other RNA viruses, such as respiratory syncytial virus, induce stress granule formation and take advantage of stress granule responses as part of the infectious cycle (58). For coronaviruses, MHV replication was found to be enhanced in cells deficient in stress granule formation, implying that stress granules contribute to viral inhibition (59)."
“All positive-strand RNA viruses that infect animals replicate in the cytoplasm of the infected host cell. However, there is ample evidence that implicates the nucleus and nuclear proteins in the replication and pathogenesis of positive-strand RNA viruses, including coronaviruses (64). The replication of these RNA viruses in enucleated cells is variable, ranging from 10% to 100% of that in nucleated controls (65, 66). The relocation of nuclear proteins to the cytoplasm and of viral proteins to the nucleus during virus replication (7, 64, 67) (Table 1) highlights the relevance of this organelle during the coronavirus infectious cycle and raises important questions: What is the role of nuclear factors in the replication of these viruses, and do viral proteins traveling to the nucleus participate in RTC activity?”
Ok, so SARS 1 can actually move to the nucleus.
“The coronavirus protein most frequently associated with the host cell nucleus is the N protein, and its transport to the nucleus is regulated by phosphorylation”
Connection of disordered part of proteins and formation of phase separation:
“Intrinsically disordered proteins (IDPs) are characterized by their biased amino acid composition and low sequence complexity and by their low content of bulky hydrophobic amino acids. Such protein sequences are unable to fold spontaneously into stable, well-defined globular three-dimensional structures but are dynamically disordered and fluctuate rapidly over an ensemble of conformations that cover a continuum of conformational space ranging from extended statistical coils to collapsed globules.”
https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4405151/
“An exciting recent finding is that many proteins containing low-complexity or prion-like sequences can promote phase separation to form membrane-less organelles within the cytoplasm or nucleoplasm, thus contributing to their compartmentalization in a regulated manner.”
I found another preprint that calls the RNA and nucleoprotein complex for inclusion body.
“Ebola virus (EBOV) inclusion bodies (IBs) are cytoplasmic sites of nucleocapsid formation and RNA replication, housing key steps in the virus life cycle that warrant further investigation. During infection IBs display dynamic properties regarding their size and location. Also, the contents of IBs must transition prior to further viral maturation, assembly and release, implying additional steps in IB function. Interestingly, expression of the viral nucleoprotein (NP) alone is sufficient for generation of IBs, indicating that it plays an important role in IB formation during infection.”
https://www.biorxiv.org/content/10.1101/2020.04.06.028423v1
rhiju, I found another interesting fact in relation to membrane-less compartments. Due to proteins in them being much more disordered, they are more accessible to post-translational modifications.
"Membrane-less compartments resulting from liquid–liquid phase separation (LLPS) are increasingly being recognized as mechanisms for organizing cellular activities."An interesting feature of proteins that undergo LLPS is they frequently contain long segments that lack well-defined three-dimensional structure. These segments are typically termed intrinsically disordered regions (IDRs), or intrinsically disordered proteins (IDPs), because they have no single equilibrium structure; instead, they exist as broad structural (or population) ensembles or they exchange between multiple conformations rapidly. IDRs are usually defined as being approximately 30 amino acids or longer [21], and their distinguishing characteristic is a relative paucity of hydrophobic amino acids to drive folding into a narrow conformational landscape.
“The lack of secondary structure makes IDRs especially susceptible to post-translational modifications (PTMs) [24]. In fact, IDRs are disproportionately modified post-translationally relative to the entire proteome [25,26,27]. A variety of PTMs can alter IDR charge, hydrophobicity, size, and structure. These changes may occur through additions of functional groups (e.g., phosphoryl, methyl, acyl, glycosyl, alkyl, etc.), or subtler chemical changes such as oxidation, deimidation, and deamidation [28].”
Also I found out there is a database over molecules found in such membrane-less compartments.
From reference 9 in the paper.
When I search that database, I can find the HDAC2 (Histone deacetylase) in there. So this is a cellular player that can move in and potentially be near nucleoprotein for modifying it.
Newly uploaded file in MobiDB. Sorted after disorder. The nucleoprotein is the protein in coronavirus judged to be the most disordered.
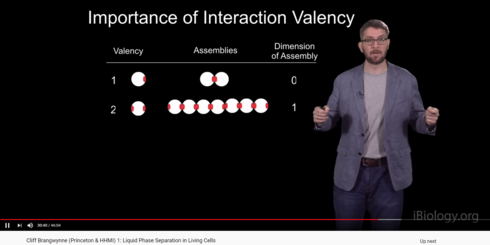
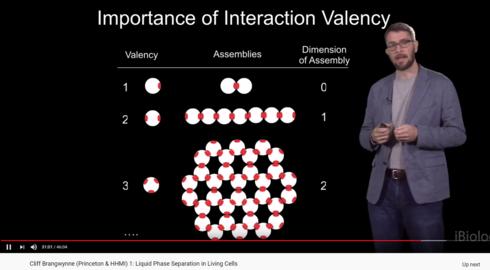
There was something in the Ibiology series on phase separation that I found very interesting.
Valency of a molecule.
Interaction of SARS and MERS Coronaviruses with the Antiviral Interferon Response
Interaction of SARS and MERS Coronaviruses with the Antiviral Interferon Response https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7112302/pdf/main.pdf
Eli- if you haven’t seen this refrence to histones you might be interested.
“…highly virulent HPAI virus and MERS-CoV induce repressive histone modifications…”
Pathogenic Influenza Viruses and Coronaviruses Utilize Similar and Contrasting Approaches To Control Interferon-Stimulated Gene Responses
eternacac, thx for the paper. I had not seen this one.
I have read a paper, forgot which one, about the new corona virus making histone modifications in the host.
So apparantly viruses making modifications to host histones seems to be a common viral feature. I find it particularly interesting that they specifically goes for dampening the host’s immune response.
Perhaps this could also help explain a bit of what Rhiju was wondering, about why the corona nucleoproteins are not binding to the host mRNA. Because there will be less of it, once the host chromatin is downreagulated. Besides that it doesn’t have the right packaging signal too.
Lots of very interesting stuff below, target tissues, histones, HDACs, and more.
A SARS-CoV-2-Human Protein-Protein Interaction Map Reveals Drug Targets and Potential Drug-Repurposing
Remdesivir disrupts RdRp complex so I propose these RNA complements be made to do the same.
Remdesivir disrupts RdRp complex so I propose these RNA complements be made to do the same.
Remdesivir disrupts RdRp complex so I propose these RNA complements be made to do the same.
Remdesivir disrupts RdRp complex so I suggest RNA complements to these contigs be tried as in picture below:

Potential small peptide disrupters of RdRp SARS-CoV-2
based on Remdesivir mechanism/action above.
RHKI
RQKI
SHKI
SQKI
RPL
SPL
RLL
SLL
TRV
TSV
I am wondering if the virus brings its own or it uses the host’s proteins. I have found some cases that involve the latter.
rhiju das:
Thought you would enjoy this –
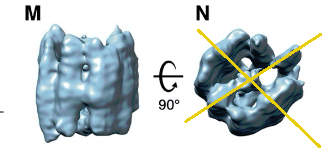
‘beads on a string’ indeed!
Eli: This is an awesome paper. I love the graphical visuals beside the photos. Ok, so the N protein is actually needed for the virus to break out of the cell. Along with the S protein.
Lol, they literally have the words bead on a string in the paper!
“We propose that vRNPs are separate complexes organized like ‘beads on a string’ that allows for efficient packing of the unusually large vRNA genome into the virus particles while maintaining high sterical flexibility between the individual vRNPs required during their incorporation into budding virions.”
I also like this bit a lot “The assembly is composed of parallel-stacked, pillar-shaped densities, presumably formed by multiple linearly aligned N proteins. Pillar-shaped densities form two densely packed curved walls opposing each other and surrounding a central density, separated from the walls by 6 nm”
Pillarshaped is a very good description
There even is some symmetry to it:
I think this paper is the closest I have ever been to seing corona virus. The N nucleoproteins are visible, but the RNA is invisible.
The N protein is really a pillar made of pillars.
So you were absolutely right. I love this paper.
Ah, the pillars on the pillars, helps with stacking the nucleoproteins on top of each other. They are ring like. Double wave stacking rings.
I think the RNA are inside the nucleoprotein. It will make no sense to have empty space inside. So I think the RNA runs like this:
So I think the pillars are the protein.
Also I wonder if the protein can be folded out like a rice mat for sushi making. So the pillars are flat.
Meaning the protein can open and attach to the RNA and then fold around it.
The RNA inside the nucleoprotein will be like a spiral staircase
Imagine sushi getting made with a rice mat.
The protein, the ricemat, the RNA, the sushi
The pillars inside the nucleoprotein are quite similar. Now I wonder if the nucleoproteins have identical sections/repeats.
rhiju das: This paper suggests that N binds to double-stranded RNA — I wonder if the sushi filling is RNA stems.
Eli: The full RNA genome of corona virus that get packaged is double stranded? I thought I read somewhere. If this is so, then yes, sushi filling is RNA stems. It also explains why proofreading can be made since there is an extra strand to correct from.
rhiju das: No only a single copy of genome
Is packaged without its complement - but it folds on itself to form lots of base pairs. Maybe those stems (hairpins and things) are bound by N to form these beads. I wish we knew!! Scientists are so behind on viruses